|
Due to repulsion between the axial F atoms and both the lone pair and double bond, we should expect the F-S-F bond angles to be compressed. For each three-dimensional molecular geometry, predict whether the bond dipoles cancel. (HCl, CH2O, NH3, and CHCl3), indicated in blue, whereas others do not because the bond dipole moments cancel (BCl3, CCl4, PF5, and SF6). With the more repulsive lone pair and the strongest equatorial repulsive force being between the double bond and lone pair, we should expect the F equatorial-I-O bond angle to be less than the 120° angle expected for the parent geometry ( it is actually much less, at 98°). Because the two CO bond dipoles in CO 2 are equal in magnitude and oriented at 180° to each other. This results in a seesaw molecular geometry. 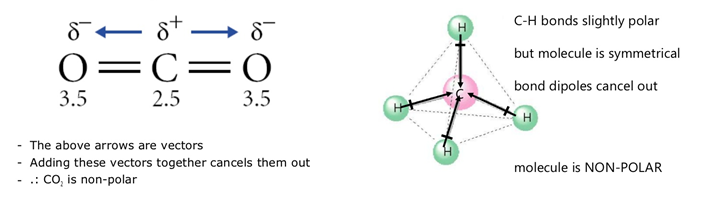
The lone pair and double bond are most repulsive, and should occupy the less crowded equatorial positions rather than the more crowded axial positions. A quick explanation of the molecular geometry of CH3OH (Methanol) including a description of the CH3OH bond angles.Looking at the CH3OH Lewis structure we ca. There is one lone pair, a double bond to O, and three single bonds to F atoms around the central I atom. This molecule has five electron groups (steric number 5) with an approximately trigonal bipyramidal electronic (parent) geometry. Happy learning Xenon Oxytetrafluoride is a colorless inorganic compound. For one bond, the bond dipole moment is determined by the difference in electronegativity between the two atoms. A dipole moment measures a separation of charge. The hybridization state for the XeOF4 molecule is sp3d2. 1: An overview of polarity and molecular shape. The molecular geometry is a distorted tetrahedron. The molecular geometry of the XeOF4 molecule is square pyramidal. As a general rule, lone pairs are slightly more repulsive than multiple bonds, and so we might expect the O-Xe-F bond angles to be 109.5° (and the actual bond angle is 120°). VSEPR theory predicts F-Xe-F bond angles of 90°. CH2O lewis structure, molecular geometry, bond angle, WebQuestion: Data. The result is a square pyramidal molecular geometry. In the CH 2 O Lewis structure, there are two single bonds around Electron. The double bond and lone pair will be directly opposite to each other, designated as axial positions. There is a double bond to O and a lone pair, both of which are more strongly repulsive than the single bonds to F. This molecule has six electron groups around the central Xe atom (steric number 6), and thus has an approximately octahdral electronic (parent) geometry.  
Use VSEPR theory to predict the geometries and draw the structures of the following.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
